Umahluko ophambili phakathi kwe-nitrate kunye ne-nitrite kukuba i-nitrate iqulethe ii-atom ezintathu zeoksijini ngelixa i-nitrite iqulethe ii-atom ezimbini zeoksijini.
Zombini i-nitrate kunye ne-nitrite i-aitrogonic adions equka i-nitrogen kunye neeathom zeoksijini. Zombini ezi aonon zinentlawulo yombane. Bebevela ikakhulu njenge-amini yeenkuku zeetyuwa. Kukho umahluko phakathi kwe-nitrate nonite; Siza kuxoxa ngaloo mahluko kweli nqaku.
Yintoni i-nitrate?
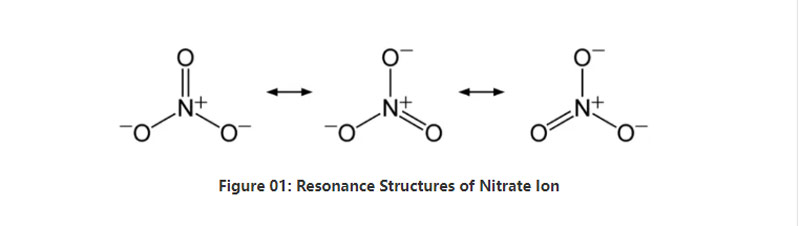
I-Nitrate yi-arion ye-inorganic yefomula yekhemikhali no3-. Yinto ye-polyatotomic eneeatom ezi-4; enye i-nitrogen Atom kunye neeathom ezintathu zeoksijini. I-ANION i -1 intlawulo. Ubunzima be-molar bale anion yi-62 g / mol. Kwakhona, le anion isuselwa kwi-acid yayo ye-Conjugit; I-nitric acid okanye i-Hno3. Ngamanye amagama, i-nitrate sisiseko se-witric acid.
Ngamafutshane, i-nitrate ion ine-atom enye ye-nitrogen kwiziko elibophelela ii-atom ezintathu zeoksijini ngokuxhobisela imichiza. Xa ujonga isakhiwo sekhemikhali sale anion, ineebhondi ezintathu ezifanayo akukho ziqhushumbisi (ngokwemibutho yokuphumla ye-ANION). Yiyo loo nto, iJomemetri yemolekyuli iludlolo lwe-trigonal. I-aptgen nganye ye-oksigen ithwala i-A-2/3 intlawulo, enika intlawulo iyonke ye-ANONI AN -1.
Kwingcinezelo esemgangathweni kunye nobushushu, phantse kuzo zonke iinqobo zazo zetyuwa ezinee-anion zinyibilikisa emanzini. Singazifumana ngokwendalo ezenzeka ngokwemvelo emhlabeni njengeeekhredithi; Iidipozithi ze-nitratini. Iqulethe i-sodium nitrate. Ngapha koko, ukongeza intsholongwane kunokuvelisa i-iite ion. Enye yezona zinto zibalulekileyo ze-timerate i-titts ikwimveliso yezichumisi. Ngapha koko, iluncedo njengearhente ye-oxidiching kwiziqhushumbisi.
Yintoni iphiko?
I-Nitite yityuwa ye-inorganic enefomula yekhemikhali no2-. Le anion yi-anion ye-vumelani, kwaye ine-nitrogen Anton i-atom ibotshwe ii-atom ezimbini zeoksijini ezifana neemichiza zekhemikhali ezinamachiza. Yiyo loo nto, iAtom ye-nitrogen iphakathi kwimolekyuli. I-ANION i -1 intlawulo.
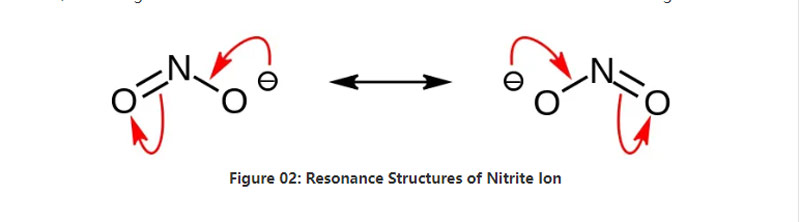
Ubunzima be-molar ye-anion yi-46.01 g / mol. Kwakhona, le anion isuselwa kwi-acid ye-nit okanye i-Hno2. Yiyo loo nto, sisiseko se-conjumetiate ye-acid ye-nit. Ke ngoko, sinokuvelisa i-nitite ye-nitite ezenziwe ngemveliso ngokudlula kwi-numes ye-zurnes kwisisombululo seShroxroxIide ye-hidroxide. Ngapha koko, oku kuvelisa iSodium Nitite esinokuyihlambulula ngokusebenzisa kwakhona i-revystallization. Ngaphaya koko, iityiwa ze-nitite ezinje ngeSodium Nitite ziluncedo kumlo wokuLondolozwa kokutya kuba zinokuthintela ukutya okukhula kwe-ronial.
Nguwuphi umahluko phakathi kwe-nitrate kunye ne-nitrite?
I-Nitrate yi-arion ye-inorganic ye-inorganing formula no3-as nitrite yityuwa ye-inorganic enefomula yekhemikhali no2-. Ke ngoko, umahluko ophambili phakathi komoya we-nitrate kunye no-nitte ubuxoki ekwenzeni iikhemizi zee-aion ezimbini. Yiyo i; Umahluko ophambili phakathi kwe-nitrate kunye ne-nitrite kukuba i-nitrate iqulethe ii-atom ezintathu zeoksijini ngelixa i-nitrite iqulethe ii-atom ezimbini zeoksijini. Ngaphezu koko, i-iiter ion isuselwa kwi-acid yayo ye-Conjugate; I-nitric acid, ngelixa i-nitite i-ion isuselwa kwi-wittcid acid. Njengomnye umahluko obalulekileyo phakathi konqulo kunye ne-nitrite, sinokuthi i-nitrate yiarhente ye-oxidiching kuba inokuphela kwakhona ngelixa i-niticing enokwenza ukuba isebenze kunye nokunciphisa iarhente.
Ixesha leposi: NgoMeyi-16 ukuya ku-2002





